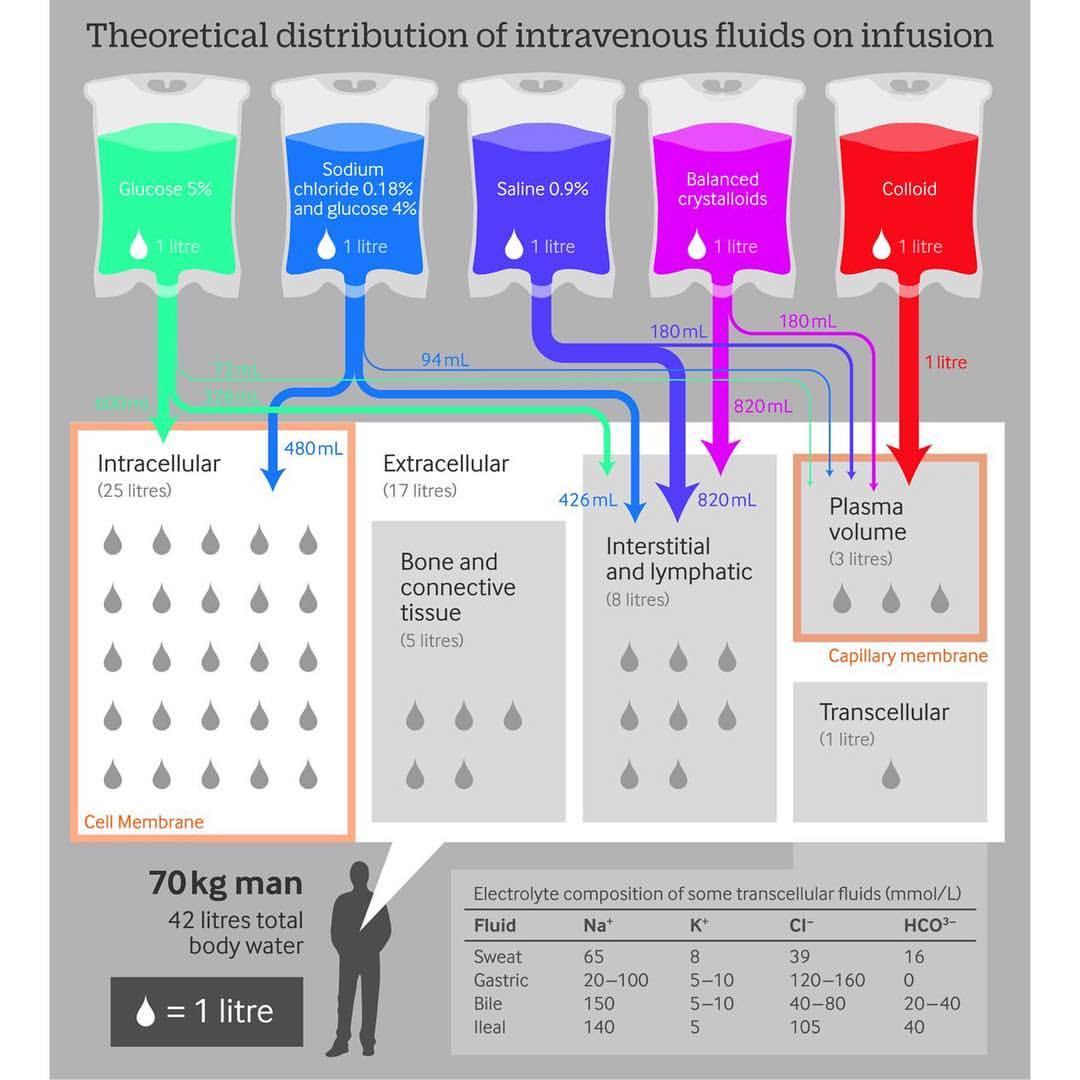
Given that the plasma volume of a person is only 3L, large amounts of body fluid and solutes are processed by the kidney each day. The glomerular filtration rate (GFR) of an average human is 180L/day. Studies have shown that sodium intake of 10 times the normal amount has relatively small changes in extracellular fluid volume and plasma sodium concentration as a result of renal compensation. The capacity of the kidneys to alter fluid excretion, as well as electrolyte excretion (e.g., sodium), is enormous. 
This fluid is reabsorbed in the renal tubes and may be modulated by hormones such as antidiuretic hormone (ADH), aldosterone, as well as angiotensin II. The kidneys play a substantial role in osmoregulation by controlling the quantity of fluid reabsorbed from the glomerular filtrate. To maintain homeostasis, the excretion of water and electrolytes must match an individual’s intake. For intracellular fluid, almost half the osmolarity is due to potassium ions, with the other half composed of various other substances (e.g., phosphate, phosphocreatine, magnesium ions). The composition of the interstitial and intravascular fluid is similar, with sodium and chloride being the primary contributors to the osmolarity. The total osmolarity for each of the three fluid compartments (intracellular, interstitial, intravascular) is around 280 mOsm/L, with intravascular being slightly greater due to the osmotic effects of plasma proteins. At low concentrations (as in the human body), these two terms are almost synonymous with each other. However, clinically, it is much easier to express body fluid quantities in liters rather than kilograms. In terms of volume status, we are concerned more so with the plasma osmolality, since it is independent of temperature and pressure. While osmolarity is the number of osmoles per liter, osmolality is the number of osmoles per kilogram. It is also noteworthy that there is a distinction between the terms osmolarity and osmolality. However, if a molecule dissociates into two ions (yielding two particles)-for instance, sodium chloride, then the 1 mol/L solution will yield an osmolarity of 2 osm/L. For instance, one mole of glucose dissolved in one liter of solution would have molarity and osmolarity of 1 osm/L (or 1 mol/L). Though similar to molarity, osmolarity refers to the total number of active particles. One osmole refers to one mole of osmotically active solute particles. The unit of osmoles is used to express the number of particles.  
The tendency of a solution to draw water in through the semipermeable membrane is the osmotic pressure. The solute concentration difference across the membrane gives rise to a gradient that facilitates the movement of a solvent (usually water in our body) until attaining equilibrium. In the human body, this selectively permeable membrane may be the cellular membrane (in the case of intracellular fluid) or maybe a membrane lining your body cavity composed of cells (in the case of extravascular fluid). Osmosis occurs when two solutions containing different concentrations of solute are divided by a selectively permeable membrane. This article will provide an overview of osmoregulation and excretion, focusing on a discussion of the renal system involved in maintaining this intimate balance between fluid retention and excretion. This process of passive regulation of osmotic pressure is known as osmoregulation. The maintenance of fluid homeostasis in each of these compartments is dependent on the excretion of fluids and the concentration of electrolytes that generate osmotic pressure. The intracellular fluid makes up roughly two-thirds of the total body volume.Įxtracellular fluid constitutes the remaining one-third of fluid volume in the body and may further divide into its interstitial, intravascular and transcellular compartments. Intracellular fluid, also considered as the cytosol, is all the fluid contained within cells. Our body fluids may subcategorize into intracellular and extracellular fluid compartments. The average adult has roughly 65% fluid mass, with this value being slightly lower in females than males. As living organisms, the maintenance of fluid balance is critical to sustaining many bodily functions, including metabolic and biochemical reactions, transport of nutrients and thermoregulation.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
